Friction
February 1, 2012
Friction in the right places can be useful. Without friction, I wouldn't be able to lift my
coffee cup without its slipping out of my hand.
Automobile travel would be impossible if the
tires just spun without grabbing the
road surface. Friction also enables the
brake pads to stop my automobile when required.
There's a dark side of friction in automobiles, and that's the reason we need
lubricating oil as well as
gasoline. Not surprisingly, only 21.5% of the
energy content of fuel actually moves an automobile powered by an
internal combustion engine.
If we look at where all the energy in fuel goes, we find that 33% is lost in
exhaust, 29% goes to
cooling and only 38% becomes
mechanical energy. Not all that mechanical energy is used to move the automobile, since 33% goes to friction losses and 5% to
air resistance.[1]
According to a study by the
VTT Technical Research Centre of Finland and
Argonne National Laboratory, friction losses in an average automobile are about 11,860
MJ per year, which corresponds to 90
US gallons of gasoline.
These losses are partitioned as follows: 35% overcoming
rolling resistance in the wheels, 35% in the engine, 15% in the
transmission and 15% in braking.[1] Since there are 612 million cars in the world, today, this adds up to 55 billion gallons of fuel wasted on friction per year.
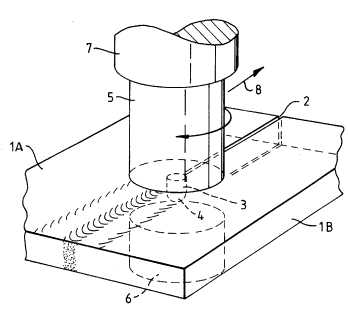
One useful application of friction in material processing.
Fig. 1 of US Patent No. 5,460,317, "Friction Welding," by Wayne M. Thomas, et al., October 24, 1995.[3]
(Via Google Patents))
VTT and Argonne state that there are technologies available to reduce friction by 10-80%, depending on the component. The benefit of rolling out these technologies would be a reduction in fuel consumption by 18% in 5-10 years, and 61% in 15-25 years. Such technologies include new
lubricant additives and
low-viscosity lubricants,
ionic liquids as lubricants and lubricant additives, surface coating and surface texturing, and tires that can be inflated to high
pressures.[1]
Diamond-like carbon and
nanocomposites are surface treatments with the potential of reducing friction by 10-50%.
Lasers can be used to pattern
microchannels on surfaces to facilitate lubricant flow and reduce friction by 25-50%. Since ionic liquids are composed of
charged molecules that repel each other, they make excellent lubricants that allow a friction reduction of 25-50%.[1]
Driver attention can help, also. If vehicle speed is reduced by 10%, fuel consumption is reduced by 16%. The report claims that reduced driving speed allows a 25% higher tire pressure, which also translates to a fuel saving; but I would be hesitant to try that for safety reasons. The analysis and recommendations were published in an article in
Tribology International.[2]
Scientists continue their quest to discover the origin of friction in particular
materials.
Graphite and
molybdenum disulfide (molybdenite,
MoS2) are two popular
solid state lubricants, and their
lubricity appears to lie in their layered
crystal structure; that is, they possess easy
slip planes. I wrote about molybdenum disulfide in a
recent article (Molybdenum Disulfide Circuitry, December 9, 2011).
Now that we have
graphene, single layers of graphite, to investigate, there are some surprises.
Scientists at the
National Institute of Standards and Technology (NIST) have used
Brownian dynamics simulation to explain the observed friction of
atomic force microscope (AFM) tips on single and multi-layer graphene sheets.[4-5]
Experiments have shown that single layers of graphene have considerable friction, but the friction is reduced in graphene stacks. The simple explanation is that the top layer of a graphene stack
deforms more when there are fewer layers beneath it.[4]
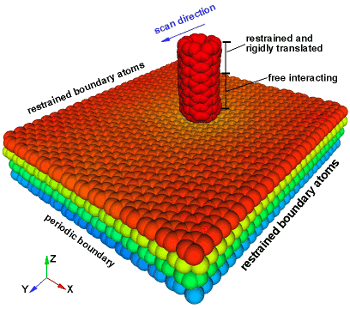
A NIST simulation of an AFM tip moving left on a four sheet stack of graphene.
Friction decreases as more layers are stacked.
(Image:
A Smolyanitsky/NIST))
The friction arises from the
deflection of graphene below and around the AFM tip. This reversible warping creates rolling resistance. When there are fewer layers, the top layer deflects more, so the friction is larger.[4]
References:
- One third of car fuel consumption is due to friction loss, VTT Press Release, January 12, 2012.
- Kenneth Holmberg, Peter Andersson and Ali Erdemir, "Global energy consumption due to friction in passenger cars," Tribology International (in press), online version, December 6, 2011.
- Wayne M. Thomas, Edward D. Nicholas, James C. Needham, Michael G. Murch, Peter Temple-Smith, Christopher J. Dawes, "Friction Welding," US Patent No. 5,460,317, October 24, 1995.
- Laura Ost, "Slippery When Stacked: NIST Theorists Quantify the Friction of Graphene," NIST Tech Beat, January 10, 2012.
- A. Smolyanitsky, J. P. Killgore and V. K. Tewary, "Effect of elastic deformation on frictional properties of few-layer graphene," Phys. Rev. B, vol. 85, no. 3 (2012), Document No. 035412 (6 pages).
Permanent Link to this article
Linked Keywords: Friction; coffee cup; automobile; tire; road surface; brake pad; lubricating oil; gasoline; energy; internal combustion engine; exhaust; cooling; mechanical energy; air resistance; VTT Technical Research Centre of Finland; Argonne National Laboratory; joule; MJ; US gallon; rolling resistance; transmission; friction stir welding; US Patent No. 5,460,317; Google Patents; lubricant additive; viscosity; ionic liquid; pressure; diamond-like carbon; nanocomposite; laser; microchannel; charge; molecule; Tribology International; material; graphite; molybdenum disulfide; molybdenum; Mo; sulfur; S; solid state; lubricity; crystal structure; slip plane; graphene; scientist; National Institute of Standards and Technology; NIST; Brownian dynamics; simulation; atomic force microscope; experiment; deformation; A Smolyanitsky; deflection.